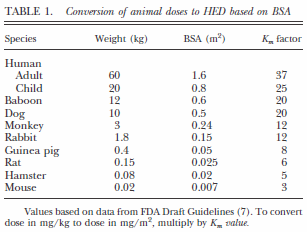
![PDF] Guidance for Industry Estimating the Maximum Safe Starting Dose in Initial Clinical Trials for Therapeutics in Adult Healthy Volunteers | Semantic Scholar PDF] Guidance for Industry Estimating the Maximum Safe Starting Dose in Initial Clinical Trials for Therapeutics in Adult Healthy Volunteers | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/8750b5d4c6564d44ac84826e3760f15ea0b836de/10-Table1-1.png)
PDF] Guidance for Industry Estimating the Maximum Safe Starting Dose in Initial Clinical Trials for Therapeutics in Adult Healthy Volunteers | Semantic Scholar
![PDF] Guidance for Industry Estimating the Maximum Safe Starting Dose in Initial Clinical Trials for Therapeutics in Adult Healthy Volunteers | Semantic Scholar PDF] Guidance for Industry Estimating the Maximum Safe Starting Dose in Initial Clinical Trials for Therapeutics in Adult Healthy Volunteers | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/8750b5d4c6564d44ac84826e3760f15ea0b836de/22-Table3-1.png)
PDF] Guidance for Industry Estimating the Maximum Safe Starting Dose in Initial Clinical Trials for Therapeutics in Adult Healthy Volunteers | Semantic Scholar
![PDF] Interpretation of Animal Dose and Human Equivalent Dose for Drug Development | Semantic Scholar PDF] Interpretation of Animal Dose and Human Equivalent Dose for Drug Development | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/f761b1fb3050f90ba97b77467f776594c3773982/5-Figure2-1.png)
PDF] Interpretation of Animal Dose and Human Equivalent Dose for Drug Development | Semantic Scholar

THE PARADOX OF HUMAN EQUIVALENT DOSE FORMULA – A CANONICAL CASE STUDY OF PIROXICAM (FELDENE) IN MONOGASTRIC ANIMALS SAGANUWAN ALHAJI SAGANUWAN (DVM, PGD, - ppt download

Characterizing the human equivalent dose of herbal medicines in animal toxicity studies - ScienceDirect
![PDF] Interpretation of Animal Dose and Human Equivalent Dose for Drug Development | Semantic Scholar PDF] Interpretation of Animal Dose and Human Equivalent Dose for Drug Development | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/f761b1fb3050f90ba97b77467f776594c3773982/3-Table1-1.png)


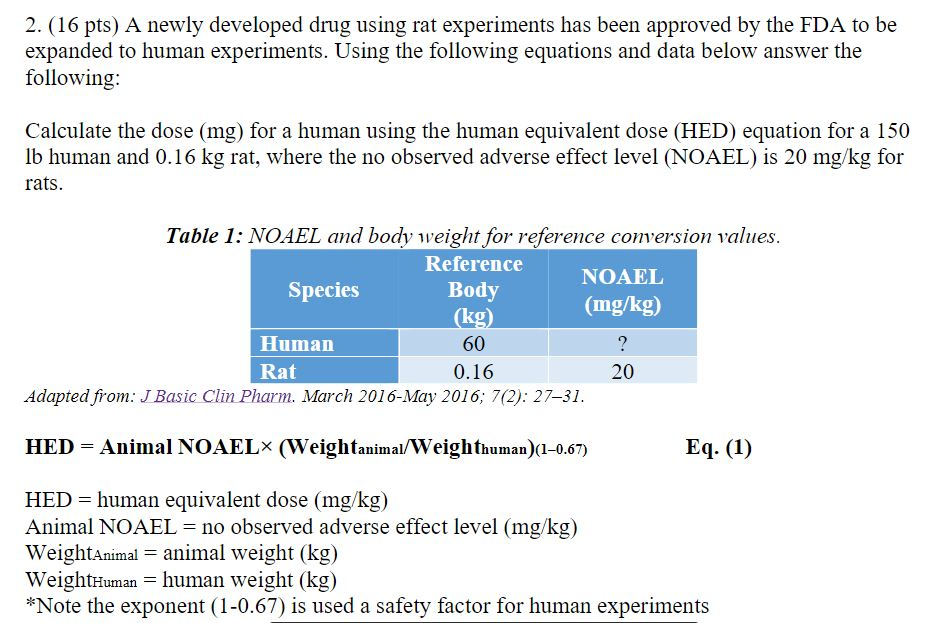




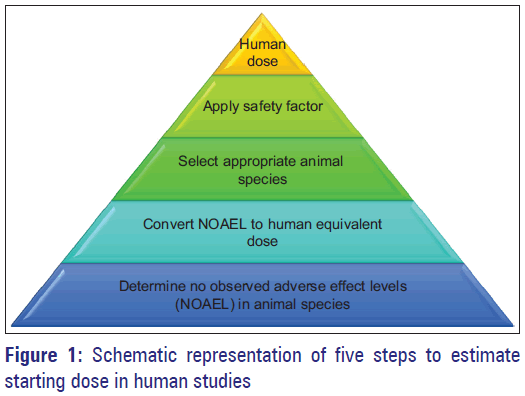



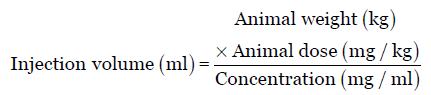
![Calculation of Equivalent Dose and Effective Dose [MOE] Calculation of Equivalent Dose and Effective Dose [MOE]](https://www.env.go.jp/en/chemi/rhm/basic-info/2018/img/img-02-03-06.png)