Guide to Irradiation and Sterilization Validation of Single-Use Bioprocess Systems - BioProcess InternationalBioProcess International

STERIS Applied Sterilization Technologies - Through locations in Europe, the United Kingdom and United States, our Radiation Technology Centers are dedicated to meeting the validation, dose audit, and research needs of our
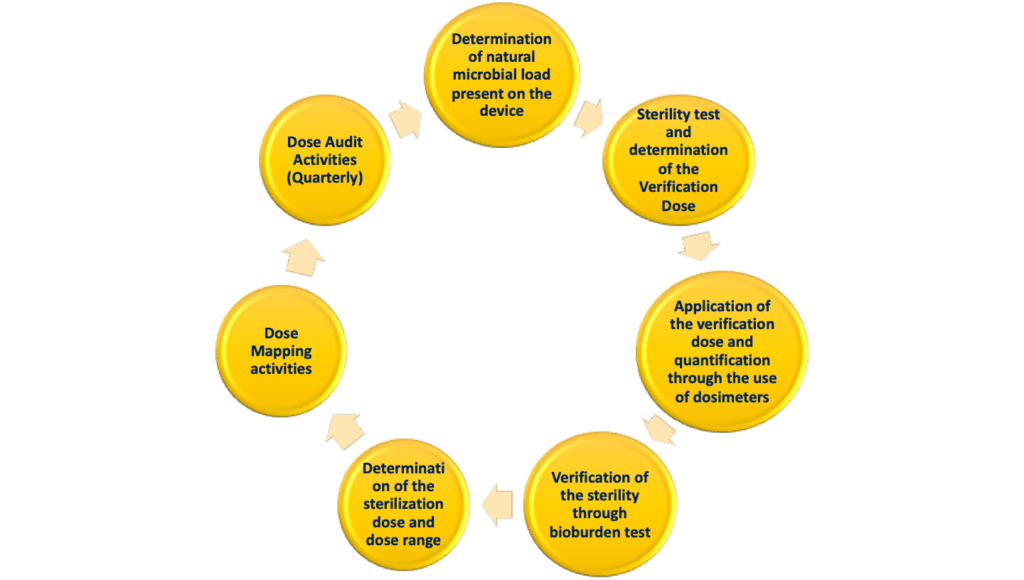
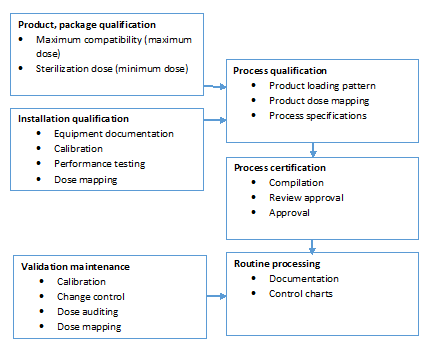
Gamma-Irradiation: Sterilization Validation Approach for Allegro™ Single-Use Systems with Sterile Claim

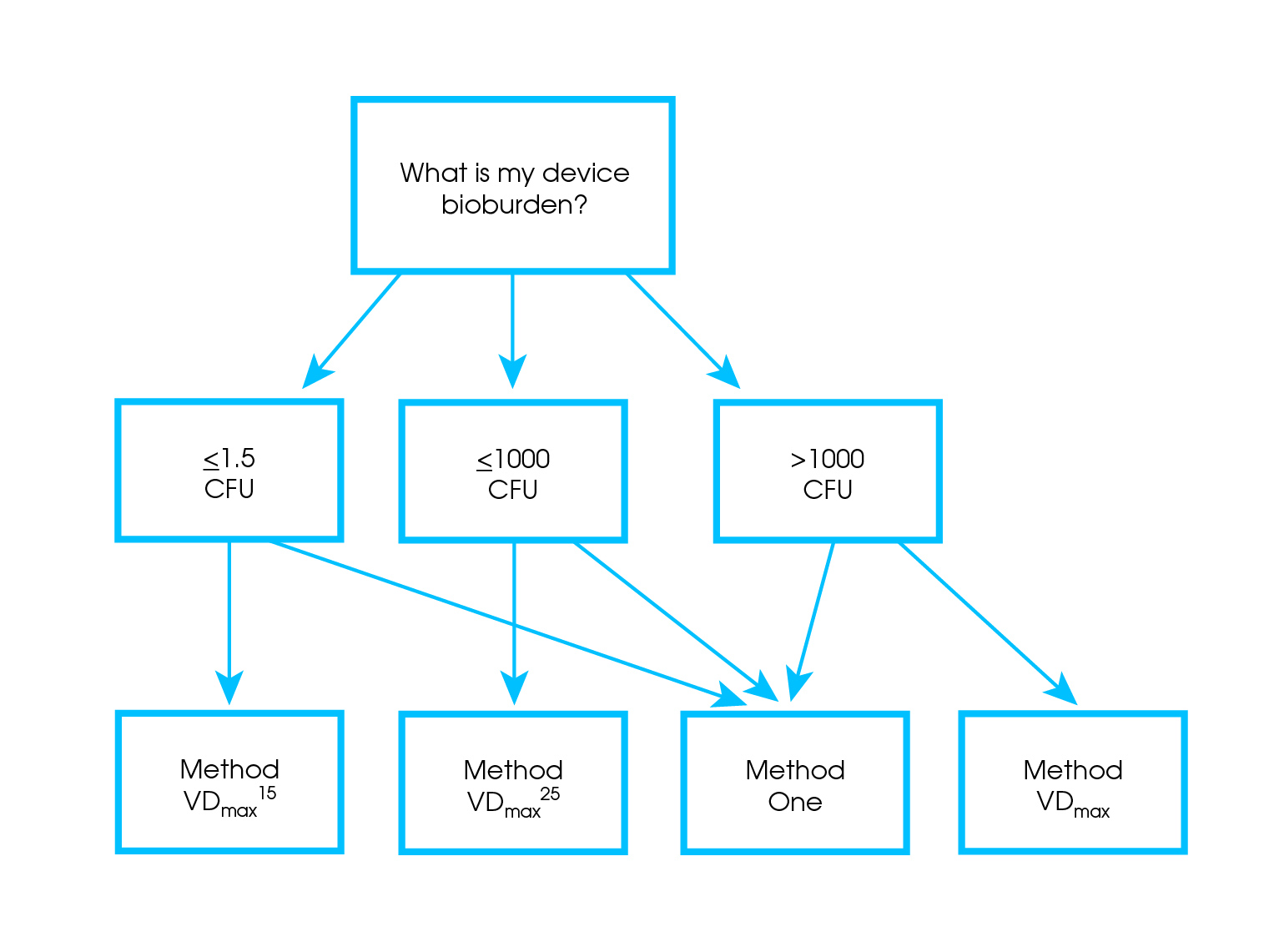
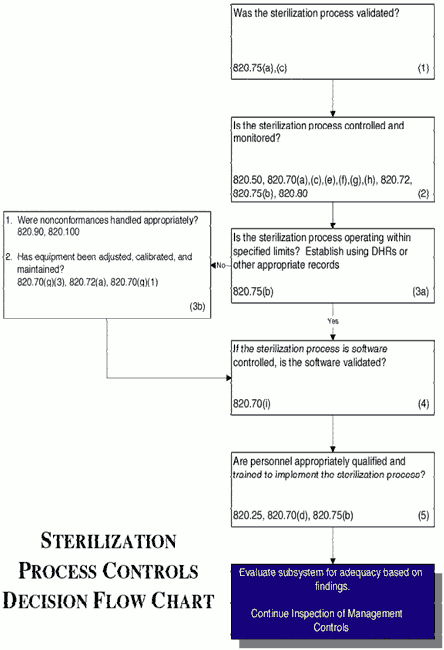
AAMI TIR35:2016 - Sterilization of health care products - Radiation sterilization - Product adoption and alternative sampling plans for verification dose experiments and sterilization dose audits.